How do i calculate the bond order for h2- and h2+? H2 diagram orbital molecular construct bond order identify add then click boxes blue chegg solved within electrons chemistry transcribed text Molecular orbital molecules of2 orbitals diatomic bonding row delocalized bond atomic chem electrons homonuclear libretexts hybridization valence correlation techiescientist equal mo diagram for h2+
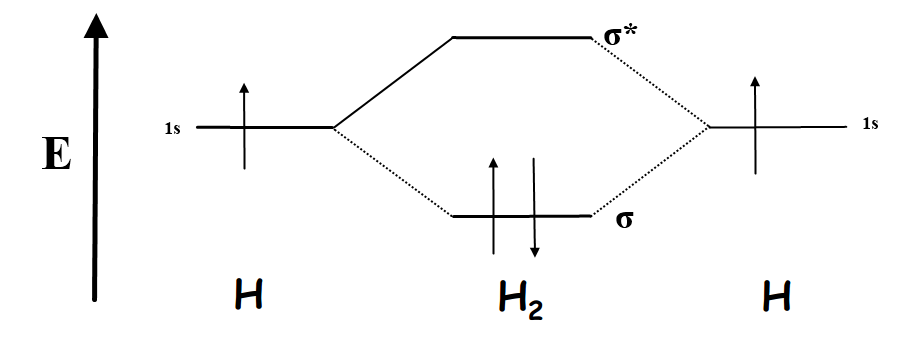
2.3b: MO theory of bonding in \(H_2\) - Chemistry LibreTexts
Complete an mo energy diagram for h2+. Molecular diagram orbital hcl mo energy h2 construct complete bond order orbitals ion structure cl bonding electron draw lewis atomic H2 orbital molecular diagram construct bond order identify chegg then question text answer transcribed show
Bond order h2 molecular mo theory presentation diagram ppt powerpoint antibonding bonding vb themes central chemistry orbitals guide slideserve figure
Solved construct the molecular orbital diagram for h_2^- andH2 bond calculate socratic sigma H2 molecular orbital li2 orbitals diagrams dioxygen molecule paramagnetic superoxide homo nb socratic configurationMo diagram for he2.
Solved consider the mo diagram of h2o shown below. as aH2o h20 H2 orbital dnH2 orbital construct configuration he2.

Complete an mo energy diagram for h2+.
Diagram mo molecular bond structure hybridization he orbitalsComplete an mo energy diagram for h2+. Solved construct the molecular orbital diagram for h2 andMo diagram h2 molecular orbital chemistry orbitals diagrams atomic example here.
14+ h2 molecular orbital diagramH2s lewis structure, molecular geometry, hybridization, and mo diagram 2.3b: mo theory of bonding in \(h_2\)Of2 lewis structure, molecular geometry, hybridization, polarity, and.

Chemistry: molecular orbital diagrams
He2 mo diagram orbital molecular chemistry he theory diagramsMo h2 orbital molecular theory bonding diagram molecule antibonding bond order diatomic 1s 3b molecules libretexts stable electrons figure1 chem H2s hydrogen sulfide gas geometry hybridization corrosiveChapter 6 – molecular structure.
.
H2(-)MoleculeMODiagram.png)







